- Blog
- Rome 2 total war faction province owner
- Gecko iphone toolkit touch 5
- Best moba games on pc
- Noteburner spotify music converter crack mac
- Iso 13485 internal audit checklist
- Flip clock screensaver windows 8
- Pokemon randomizer fire red seed
- Project budget plan template
- Manycams old version
- Ufc 3 pc download ocean of games
- Solve a quadratic equation using square roots
- Mac os disappered in boot camp
- Red giant shooter suite serial
- Divine feminine paintings
- Ptsd diagnostic criteria for dsm 5
- Keno numbers mass
- Wii u guitar hero live guitar controller
- Github desktop pull from fork
- Nate newton wiki
- Dirt 3 product key generator
- Cool house designs for terraria
- Robotc commands list robotc natural language pltw
- Resource packs for minecraft xray
- Minecraft xray resource pack 1-10-2
- Bmi calculator women for wt loss
- Nsfw ffxiv discord
- Fietype pdf learn robotc
- Vegas pro 14 serial number only numbers
- Gb whatsapp app download apk
- Patching lath and plaster walls
- Stanley sharpshooter staple gun not working
- Soulcalibur iv raphael
- Best monthly budget planner excel
- Tall sprite clothes rpg maker mv
- Free flow charts templates word
- Free download daily schedule template for kids
- Online battleship game free
- Loan arrangement fees accounting treatment
- Download cinema 4d r19 studio free
- Us mail forwarding service international
- Funny fantasy football team names ezekiel elliott
- Test internet connection speed
- Tv channels list for spectrum stream
- Quest diagnostics appointment check
- Sakura dungeon lewd patch
- Office 2010 64 bit download gratis
- Xenia xbox 360 emulator xenia download
- Solving a quadratic equation
- Avery label 5167 template google doc
For this purpose, the checklist contains the basic audit questions for all standard requirements of the current ISO 13485:2016 and supports companies in meeting the individual requirements.
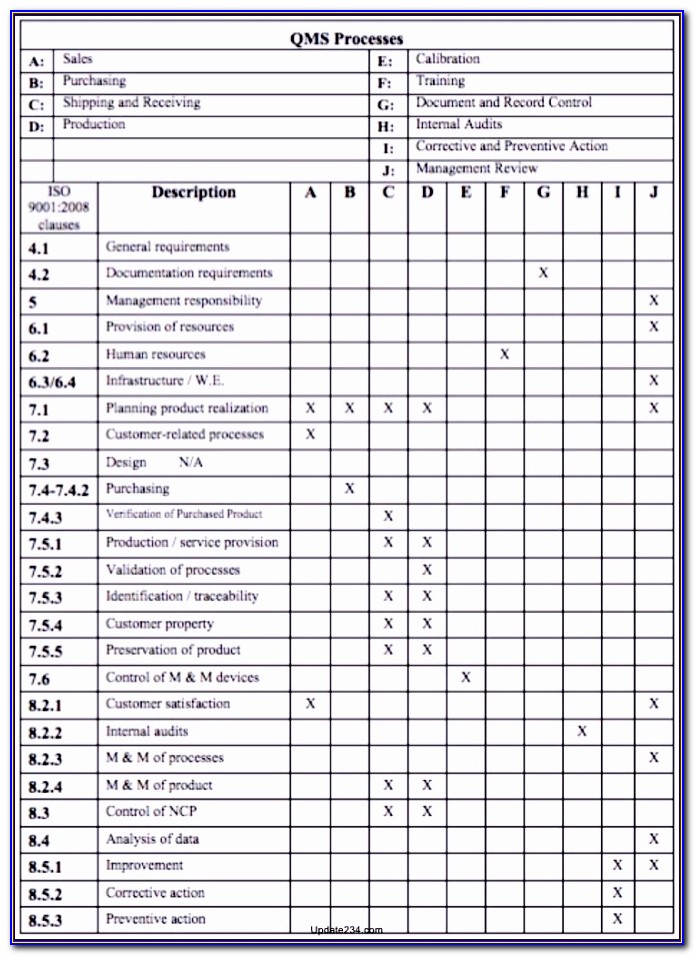
The template should be structured in such a way that all areas can be processed step by step without omitting important aspects. The audit checklist should include all relevant standard requirements of ISO 13485 and, in the best case, additionally the provisions of the Medical Devices Regulation 2017/745/EU (MDR) and/or the Medical Devices Directive (93/42/EEC). The best way to do this is with an ISO 13485 audit checklist, which should be designed as a questionnaire for the professional conduct of an audit. With an internal ISO 13485 audit, medical device manufacturers and providers prepare for external certification of their QM system. gain competitive advantage through independent certification.communicate their high quality standards.companies confirm that they meet the aspects of the requirements of relevant guidelines for medical devices covered by the standard.With certification to EN ISO 13485 by an independent certification body:
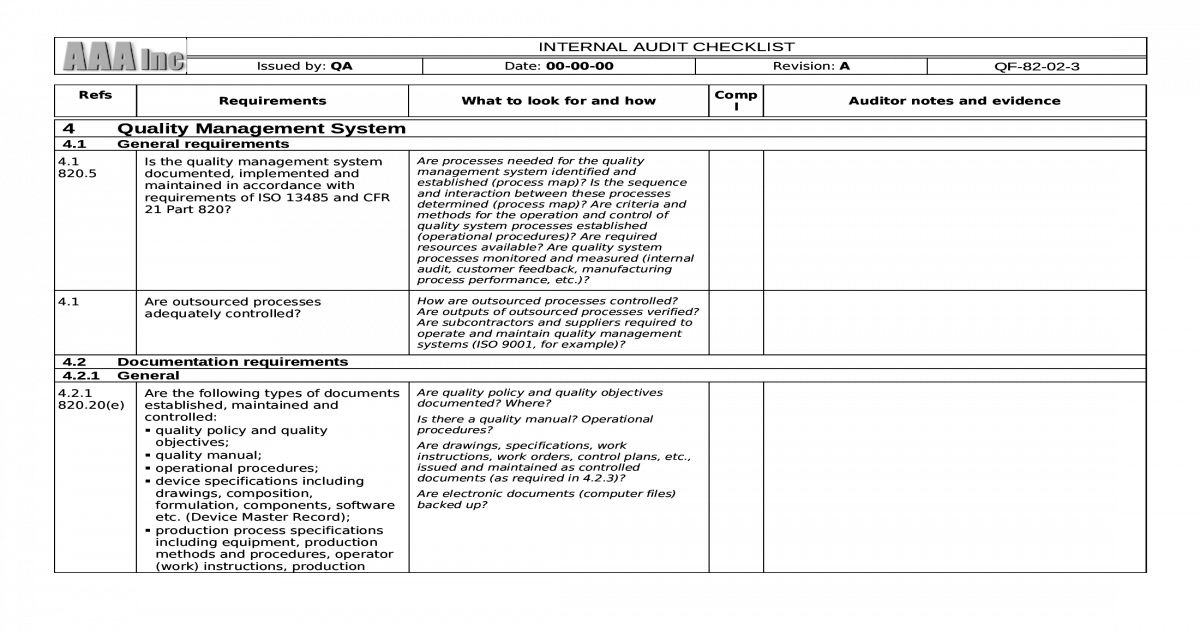
This can lead to delays in market entry and, consequently, to lost sales. Medical devices manufactured or offered by companies that do not have a certified ISO 13485 quality management system are usually denied access to important markets. App & software for obtaining ISO 13485:2016 certification The preparation for an ISO 13485:2016 certification auditģ. The relevance of ISO 13485 certificationĢ. However, such a checklist can also be used by internal quality management teams to prepare for the external ISO 13485 audit.ġ. Using an ISO 13485 checklist, external inpectorsĭetermine whether a company’s QM system complies with the standard’s requirements and is thus suitable for certification. However, it contains additional requirements for medical devices and reformulates some of the requirements of ISO 9001 for its own purposes. In substance, ISO 13485 is similar to the ISO 9001 standard. In addition to QMS requirements, the standard also includes customer requirements and the regulatory requirements of the European Union (EU), Canada, and other major markets worldwide. For this reason, national and international regulatory agencies require medical device manufacturers and providers to demonstrate that they have a quality management system (QMS) that has been audited and certified by an external body.Ĭertification bodies generally base their audits on the EN ISO 13485 standard “Medical devices: Quality management systems – Requirements for regulatory purposes.” This addresses the requirements that medical device manufacturers and suppliers must meet in developing, implementing, and maintaining quality management systems. The quality and effectiveness of medical devices affect the safety of patients, users and others more than many other products. It is critical to ensure that the problems found during the audit were addressed and respective action is taken to resolve them.Why use checklist to audit for ISO 13485 certification? Corrective action and Preventive action (CAPA) are an integral part of the audit process.įollow-up is an essential part of a successful audit. If in the audit any non-conformities or issues are identified, it is crucial to take action to resolve them and ensure that they are not repeated in the future.
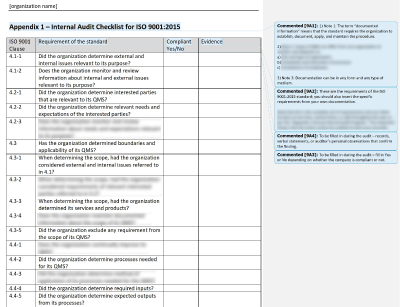
The auditor should provide a written report summarizing the audit results and any proposed corrective action. Post-audit, the auditor will meet with the stakeholders to discuss the findings and also provide any potential improvements that could be made. This depends totally on the organization and management.Īfter deciding when the audit is to be conducted, a concrete plan is required to ensure the availability of all the stakeholders on the scheduled date and time.Īn audit may consist of reviewing past records, interviewing employees, or observing the process itself. Internal audits are typically conducted twice a year or annually. This step must clearly define what is to be audited, who is responsible for the audit, and when the audit should be done. We have broken down the requirements for better understanding:Īn organization’s internal audit program must include documentation and all implementation components. The internal audit section in 13485 mentions that the organization performs an internal audit program, solves problems identified during the audit, and verifies if problems are resolved. Any medical device manufacturing company is required to carry out the ISO 13485 internal audits regularly to ensure the quality and safety of the device.
